China plans to test a recombinant vaccine against coronavirus in Uzbekistan
On October 21, the Ministry of Innovation and Development held talks with representatives of the Institute of Microbiology of the Chinese Academy of Sciences and the Chinese company Anhui Zhifei Longcom Biopharmaceutical on the organization of clinical trials of the recombinant vaccine against coronavirus in Uzbekistan.
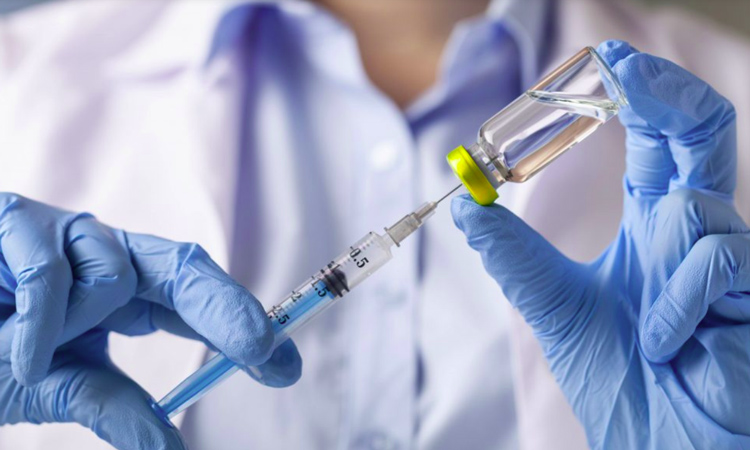
The vaccine developed by the company and currently undergoing clinical trials is a genetically engineered protein of the surface antigen coronavirus, with a high degree of safety compared to adenovirus and inactivated types of vaccines, the Ministry informed.
“During the negotiations, it was noted that in case of successful completion of all phases of clinical trials, Uzbekistan will have priority in acquiring a vaccine at a minimum price,” the report says.
Specialists from the Ministry of Innovation Development discussed the organization of the vaccination process with Chinese partners and preparations for it. Clinical trials are expected to be conducted on volunteers aged 18 years and older. Before the trials begin, representatives of the People’s Republic of China will conduct a series of trainings on vaccination conditions.
Related News

18:30 / 04.06.2026
Uzbekistan and Hong Kong eye fintech, green energy, and direct flights in new partnership

15:22 / 02.06.2026
China and Russia remain Uzbekistan’s top trading partners as FTT hits $26.3 billion in four months

16:21 / 29.05.2026
China's Spring Airlines may launch direct flights to Uzbekistan

11:50 / 29.05.2026




